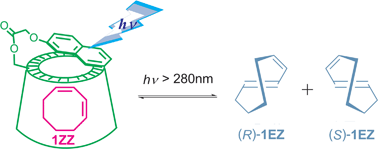
Three naphthalene-modified cyclodextrins (CDs) 3–5 were synthesized as supramolecular chiral photosensitizing hosts for enantiodifferentiating photoisomerization of (Z,Z)-1,3-cyclooctadiene (1ZZ) to its E,Z-isomer (1EZ). In aqueous methanolic solutions, β-CD-based sensitizer 4 binds 1ZZ in its chiral cavity much more strongly than α- and γ-CD homologues 3 and 5. Accelerated, often static, fluorescence quenching of these naphthalene-modified CDs was observed upon inclusion of 1ZZ. The photoisomerization of 1ZZ mediated by 3–5 yielded enantiomeric 1EZ in modest yields. The enantiomeric excesses (ee’s) obtained with α- and β-CD-based sensitizers 3 and 4, both of which have relatively small cavities, are less sensitive to temperature, demonstrating the low-entropy nature of the α- and β-CD complexes. In contrast, increasing reaction temperature significantly diminished the product’s ee and even caused a switching of enantioselectivity upon photoisomerization sensitized by γ-CD-based 5, revealing that the entropy factor plays a crucial role in the wide cavity of γ-CD.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...